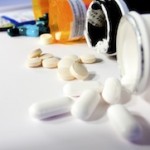
Over the last 24 months, more than 14 million units of drugs and medical equipment have been recalled due to defect. The latest was a February 17th announcement by the makers of Tylenol who recalled 574,000 bottles of infants’ Tylenol Oral Suspension. The recall is due to the product’s simple dosing measuring system that may fail and fall in to the suspension. Although … [Read more...] about What’s With All of The Recalls? Latest Infants Tylenol Recall and More
Financial Literacy: What to Teach Younger Children
Financial literacy is an important part of raising children to be successful and independent. However, good money habits don’t just develop spontaneously. Children need to have instruction in financial literacy from the time they are small. It is possible for you to teach younger children about money, building a solid financial foundation for them in the years that … [Read more...] about Financial Literacy: What to Teach Younger Children
Superbowl Weekend and Links
As a New Yorker I'm stoked to see the Giants in the Big Game! It will be fun to watch it with the kids too. Though I suspect their interest will wane pretty quickly. That's OK, the little ones are still young (our oldest daughter isn't that interested in it). Do you get the whole family involved in the SuperBowl? Do you hold a big party? How do you get everyone … [Read more...] about Superbowl Weekend and Links
9 Inexpensive Ways to Amuse Kids Over the Summer
Summer vacation has arrived. For many of us, that means the constant refrain of, “I’m bored!” You want to provide something for your kids to do (other than watch TV or play video games), but you don’t want to spend a lot of money. Happily, there are some inexpensive things that you can do to amuse your kids over the summer. 9 Inexpensive Ways to Amuse Kids Over the … [Read more...] about 9 Inexpensive Ways to Amuse Kids Over the Summer
Festival of Frugality #290 – Going on Vacation
It is our absolute pleasure to be hosting the Festival of Frugality this week. As you know, the Festival of Frugality contains the best articles on frugality and saving money from sites big and small. This week we're going on vacation so I thought we'd share some of our tips on going away. Let's get right to the frugal awesomeness! These are the Editor's Picks: … [Read more...] about Festival of Frugality #290 – Going on Vacation